In this blog, we will share our spotlight on Caplin Point Laboratories Ltd.
Caplin Point Laboratories Ltd. – Highlights
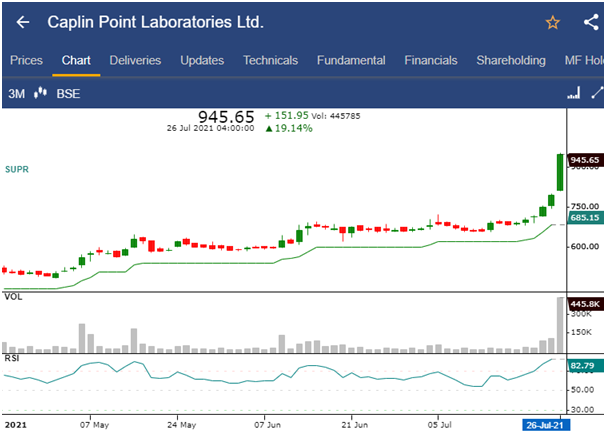
- The shares of Caplin Point Laboratories Ltd. was trading at Rs.945.65, up by 19.14%, hitting a 52 week high of Rs.952.40 in today’s trading session.
- The stock price has increased by 34.73% in last one week, while the benchmark index S&P BSE Sensex has increased by 0.57%.
- The stock has surged 37% in the last 7 trading days after after its subsidiary Caplin Steriles received approval from the United States Food and Drug Administration (USFDA) for generic Sumatriptan injection used for acute treatment of migraine and cluster headache.
- “The United States Food and Drug Administration (USFDA) has granted final approval to Caplin Steriles for its Abbreviated New Drug Application (ANDA) Sumatriptan Injection USP, 6 mg/0.5 mL (12 mg/mL) single-dose vials. Caplin Point Laboratories said in a press release that the product is a generic therapeutic equivalent version of GlaxoSmithKline’s (RLD), IMITREX Injection.
- Sumatriptan Injection USP is approved for the acute treatment of migraine with or without aura, as well as the acute treatment of cluster headache in adults. According to IQVIATM, Sumatriptan Injection had US sales of approximately $5 million for the 12-month period ending March 2021 for vial presentation (IMS Health).
- Caplin Point Laboratories Ltd’s Board of Directors, at its meeting on May 6, 2021, declared an Interim Dividend of Rs. 1.50/- (75%) per equity share of Rs. 2 each for the fiscal year ended March 31, 2021.
- Caplin Steriles Ltd, a subsidiary of Caplin Point Laboratories Limited, is a sterile product manufacturing company that is FDA and EU-GMP approved. Caplin Steriles Limited has developed and filed 20 ANDAs in the United States on its own and with partners, with 16 approvals so far. The company is also working on a portfolio of 35 simple and complex injectable and ophthalmic products that it plans to file over the next four years.
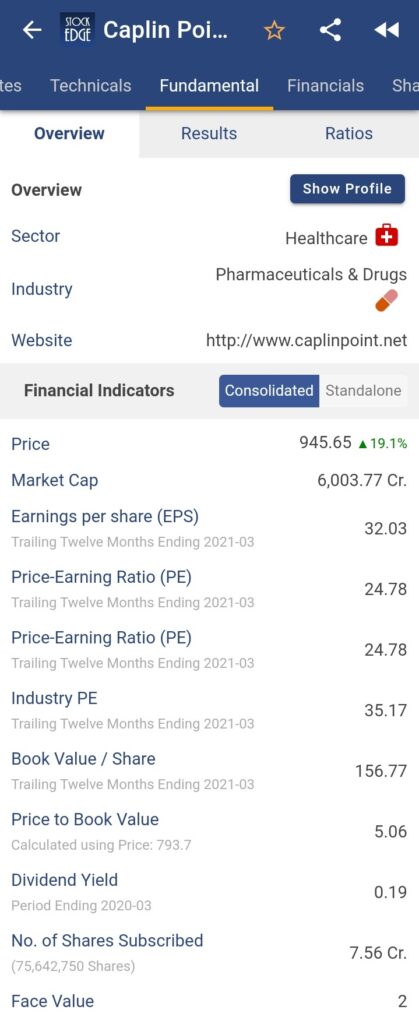
- Caplin Point Laboratories Limited is a rapidly expanding pharmaceutical company with a distinct business model focused primarily on emerging markets in Latin America and Africa. Caplin Point has cutting-edge manufacturing facilities that can produce a wide range of finished dosage forms. Caplin Steriles Limited, a subsidiary of the company, serves the Regulated Markets for Injectable and Ophthalmic products.
For more fundamental data and analysis, click on Caplin Point Laboratories Ltd.
Read our latest article on Just Dial – Why Reliance Retail acquired a stake in it?
To get more detailed analysis and Reports on Stocks, visit our Edge Report Section by subscribing to our StockEdge premium plans
Disclaimer: This document and the process of identifying the potential of a company has been produced for only learning purposes. Since equity involves individual judgments, this analysis should be used for only learning enhancements and cannot be considered to be a recommendation on any stock or sector.








Many thanks for this article. It is informative and useful.