Check out the share trend of Bajaj Healthcare Ltd.
Bajaj Healthcare Ltd. – Highlights
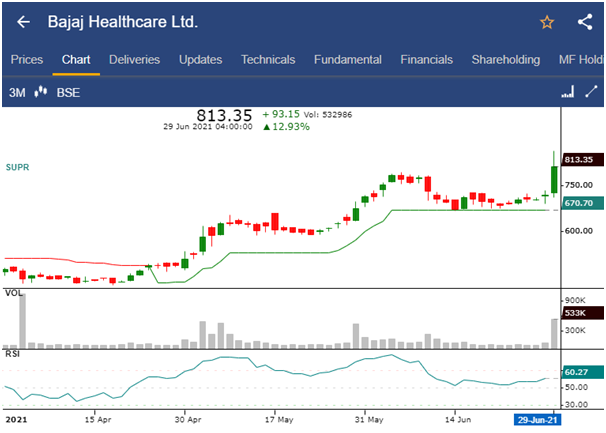
- The share price of Bajaj Healthcare Ltd., was trading at Rs.813.35, up by 12.9%, hitting a 52-week high of Rs.863.95 in today’s trading session.
- The stock price has increased by 17.77% in last one week, while the benchmark index S&P BSE Sensex has decreased by 0.07%.
- The stock gained after the company announced that it had applied to the Indian Patent Office requesting for a compulsory licence to manufacture and supply the Covid-19 drug “Baricitinib” (API and formulation).
- The US Food and Drug Administration (FDA) has granted Eli Lilly and Company an Emergency Use Authorization (EUA) for the distribution and emergency use of “Baricitinib” in hospitalised adult and paediatric patients aged more than two years with suspected or laboratory confirmed COVID-19 who require supplemental oxygen, invasive mechanical ventilation, or extracorporeal membrane oxygenation (ECMO).
- The EUA approves the use of baricitinib in combination with remdesivir for the treatment of suspected or laboratory confirmed COVID-19 in hospitalized adults and paediatric patients over the age of two who require supplemental oxygen, invasive mechanical ventilation, or ECMO.
- The company approached Eli Lilly & Company twice to sign a voluntary licence for the manufacture and supply of the Covid-19 drug baricitinib. BHL assured them that it would manufacture high-quality products at a reasonable cost for the Indian patient population. Eli Lilly & Company declined the applications on both occasions, citing the difficulty in signing voluntary licencing agreements with all of the Indian companies who had requested them.
- The company said, “When all attempts to obtain a voluntary licence from the Patentee proved futile, BHL approached the Indian Patent Office, requesting a compulsory licence for the manufacture and supply of Covid-19 Drug Baricitinib, so that it could supply the essential medicine in India during this pandemic”.
- The company recently announced the launch of APIs and formulations for ‘Favipiravir,’ an antiviral, ‘Ivermectin,’ an anti-parasitic drug used to treat mild to moderate Covid 19 disease, and Posaconazole API, a triazole antifungal agent used to treat Mucormycosis (Black Fungus) infection in Covid-19 patients.
- Bajaj Healthcare Ltd., founded in 1993, is a bulk drug manufacturer dedicated to serving the pharmaceutical, nutraceutical, and food industries globally. In 1993, the company began operations with a single unit in Tarapur. Bajaj Healthcare went public in 2005 and its shares were listed on Indian stock exchanges in 2016. The company has four APIs/intermediates units and one formulation unit that are designed to meet the needs of both advanced and emerging market opportunities.
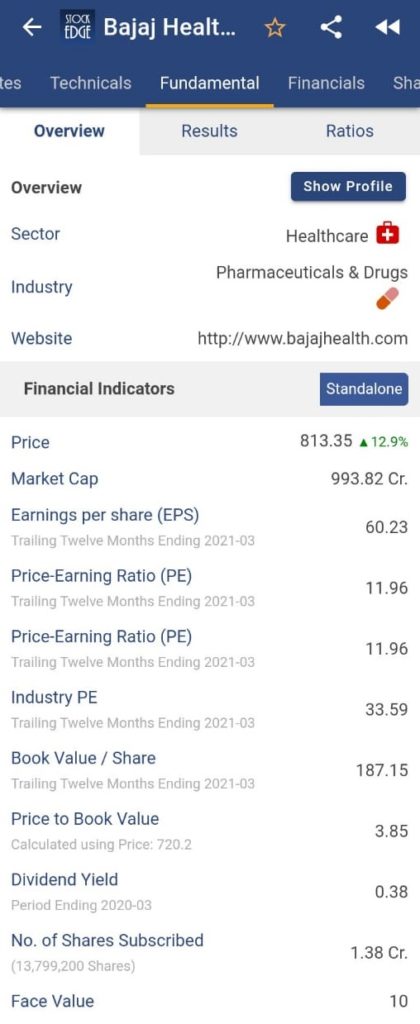
For more fundamental data and analysis, click on Bajaj Healthcare Ltd.
Read our latest article on Laurus Labs Ltd. – A leading research-driven pharmaceutical company
To get more detailed analysis and Reports on Stocks, visit our Edge Report Section by subscribing to our StockEdge premium plans
Disclaimer: This document and the process of identifying the potential of a company have been produced for only learning purposes. Since equity involves individual judgments, this analysis should be used for only learning enhancements and cannot be considered to be a recommendation on any stock or sector.